|
7/5/2023 0 Comments Density formula 
The intake condition of the propellants could affect their rate of reaction somewhat, causing the engine to have a different optimal O/F ratio. The formula for density is: m V m V where is density measured in kilograms per cubic meter, m is the mass of the objects measured in kilograms, and V is the volume of the object. You can't pump solids, another engineering concern.Īnd would T and P other than standard conditions affect the O/F ratio, rw? A small boost in kerosene density can be achieved by chilling it down slightly, but this is limited by the freezing point of kerosene. To compress the liquid for a very slightly better density, the container must be much stronger and heavier. In this case, then what is the density of the fuel? Meaning, how do we select the P and T at which the density of the kero should be determined? 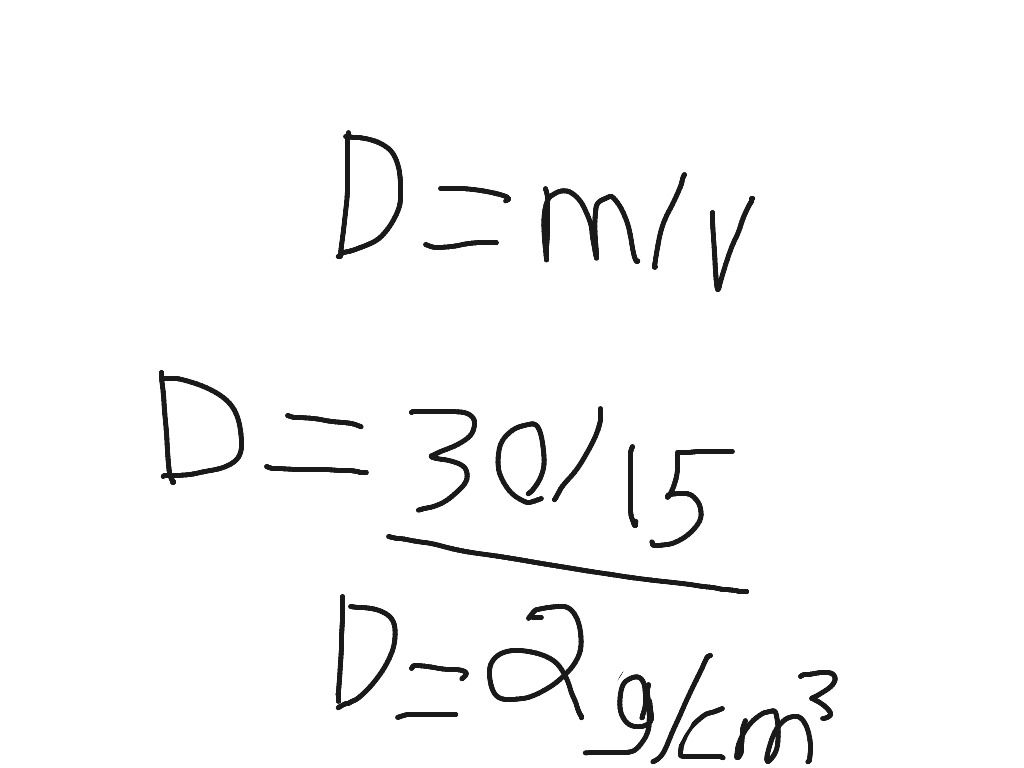
Instead low temperatures are used, typically a little below the boiling point in order to be able to have the rocket stand by for a little while before the propellant boils off. 
So the pressure is close to atmospheric for practical reasons. If you need to calculate the density of an object manually, use the density formula. This formula can be used to determine the density of any substance. As there are many units of mass and volume covering many different magnitudes there are a large number of units for mass density in use. That would cause the dry weight of the rocket to be much higher, decreasing performance. Density is calculated according to the simple formula: pM/v That is, density (p) is equal to total mass (M) divided by total volume (v). Density calculation has been changed: equation 25 in Chengs paper to compute the density of the mixture should use the glycerine fraction by VOLUME and not. From the equation for density ( m / V ), mass density has any unit that is mass divided by volume. So what two things do we need to determine density Mass Volume. High pressure is impractical, since pressure vessels are heavy. Density The average density of a substance or object is defined as its mass per unit volume, m V m V where the Greek letter (rho) is the symbol for density, m is the mass, and V is the volume. The formula is listed above: mass divided by volume. However, this needs to be at a pressure higher than atmospheric pressure, correct? Or at a very low temp. What would the bulk density of the LOX be? I have so far assumed 1,141 kg/L. Calculating density using grams and cubic centimetres would give a density unit of grams per cubic centimetre (g/cm³). In general, liquids have rather small variations in density, so standard reference values are usually good enough for a wide range of rocket configurations. To address the part about temperature and pressure, these are engineering considerations, based on what can practically be achieved. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
